Product Description
| Download Documents | |
| SDS | Product Sheet |
| COA_CM02025.1_Buffer A_0102E COA_CM02025.1_S651.S2.0416F |
|
| COA_CM02025.1_Buffer B_0102E COA_CM02025.2_S651.S2.0416F |
|
Hydrophobic Interaction Chromatography (HIC) Buffer Set
We are pleased to introduce a new HIC buffer format featuring significantly reduced cost, enhanced formulation with extended stability, ambient storage, increased quantity per unit, lower shipping costs, and user-friendly design improvements.
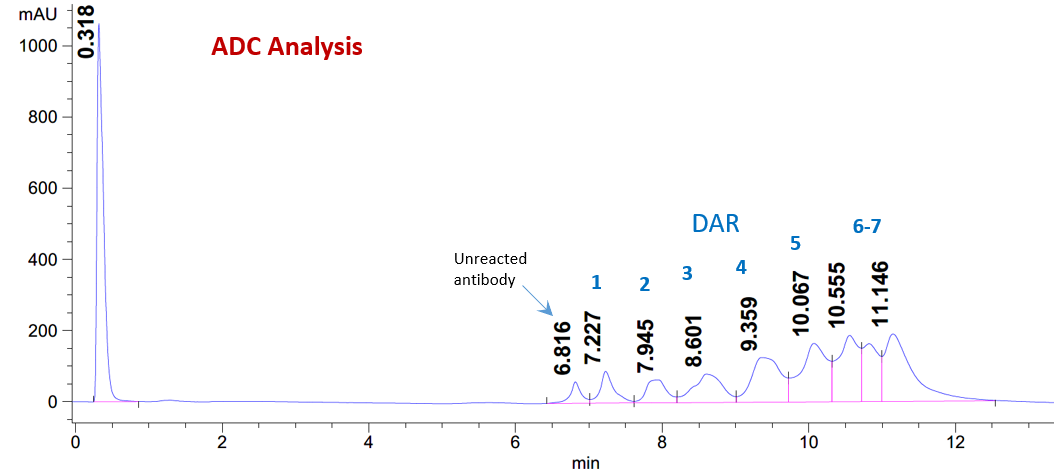
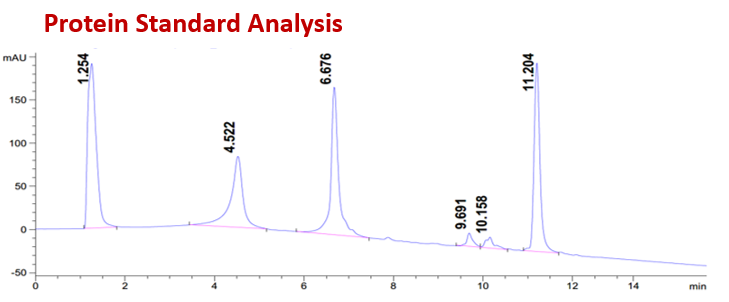
CellMosaic’s HIC HPLC Analysis Buffer Set is designed for the convenient separation and analysis of bioconjugates labeled with highly hydrophobic small molecules based on differences in hydrophobicity. Hydrophobic interaction chromatography (HIC) is a useful technique for characterizing bioconjugates such as antibody-drug conjugates (ADCs) as well as protein- or peptide-drug conjugates. The interaction between the bioconjugates and the column media is influenced by the salt concentration or organic solvent composition of the running buffer.
To support bioconjugate analysis, CellMosaic has developed this high-quality HIC buffer set for use following preparation of bioconjugates with CellMosaic’s Personalized Conjugation Kit™ (PerKit™).
A high salt concentration promotes stronger interaction between the bioconjugates and the column media. In a typical HIC method, samples are eluted using a decreasing salt gradient. Under these conditions, bioconjugates with the lowest degree of labeling and therefore lower hydrophobicity elute first, while more highly labeled, more hydrophobic bioconjugates elute later. If the conjugated molecule is hydrophilic, such as CellMosaic’s proprietary AqT® molecules, the elution trend is reversed. In this case, bioconjugates with a higher degree of labeling become more hydrophilic and exhibit weaker interaction with the HIC column. As a result, highly labeled species elute earlier, while less labeled, relatively more hydrophobic species elute later under a decreasing salt gradient.
Key-Features
- Experimentally selected ultra-pure components ensure minimal UV background absorbance.
- Compatible with all standard HIC columns for biopolymer analysis.
- Each buffer set provides sufficient volume for 120 analyses, depending on usage conditions,
- All buffer bottles and components are sterile.
Application of the product
Antibody-Drug Conjugates (ADCs) & Bioconjugates
DAR determination, conjugation heterogeneity, species separation.
Protein Purification (Preparative HIC)
Capture and polishing in downstream processing.
Protein Variant and Isoform Separation
Separation of isoforms and subtle structural variants.
Protein Folding and Conformation Studies
Monitoring folding/unfolding and conformational changes.
Aggregate Analysis
Detection of hydrophobic aggregates.
Viral Vector and Vaccine Purification
Purification of viral particles and vaccine proteins.
Peptide and Protein-Drug Conjugates
Analysis of peptide-drug and protein-small molecule conjugates.
Biosimilar and Biologic Comparability
Comparing originator vs biosimilar products.
Quality Control in Biopharmaceutical Manufacturing
Process monitoring and batch consistency.
Enzyme Purification and Characterization
Isolation of enzymes and isoenzymes.
Components
The HIC buffer set includes two buffers and instructions for use (120 injections, depending on use):
| Name | Part # | Volume upon dissolution | Description |
| HIC Buffer A | CM02025.1-1000ML | 1 Liter | 0.1M Sodium phosphate buffer, 1.8 M Ammonium sulfate, pH 7.0 ± 0.1 |
| HIC Buffer B | CM02025.2-1000ML | 1 Liter | 0.1M Sodium phosphate buffer, pH 7.0 ± 0.1 |
Supplied as powder in two bottles. Dissolve in HPLC-grade deionized water prior to use.
Storage and Solution Stability
Undissolved HIC Buffer A and Buffer B are stable at ambient temperature for up to 5 years.
Reconstituted HIC Buffer A should be stored at room temperature and is stable for up to 1 year. Do not store at 2–8 °C to avoid salt crystal formation.
Reconstituted HIC Buffer B can be stored at ambient temperature for up to 1 week. For longer-term storage, store at 2–8 °C to prevent microbial or algae growth.
Key References
Antibody-Drug Conjugates (ADCs) & Bioconjugates:
Chen, X. et al., mAbs, 2016, 8(7): 1210–1223.
Highlight: Demonstrates that HIC enables high-resolution separation of ADC species by DAR, making it a primary analytical method for ADC characterization.
Protein Purification (Preparative HIC):
Queiroz, J.A. et al., Journal of Biotechnology, 2001, 87(2): 143–159.
Highlight: Shows HIC as a mild, non-denaturing purification technique that preserves protein structure and achieves high recovery.
Protein Variant and Isoform Separation:
Fekete, S. et al., Journal of Pharmaceutical and Biomedical Analysis, 2014, 101: 161–173.
Highlight: Illustrates that HIC can resolve monoclonal antibody variants based on subtle hydrophobic differences, complementing other analytical methods.
Protein Folding and Conformation Studies:
Fausnaugh, J.L., Kennedy, L.A., Regnier, F.E., Journal of Chromatography A, 1984, 317: 141–155.
Highlight: Shows that unfolded or partially folded proteins exhibit stronger retention due to exposed hydrophobic regions.
Aggregate Analysis:
Arakawa, T. et al., Protein Science, 2006, 15(6): 1487–1495.
Highlight: Highlights HIC’s ability to detect hydrophobic aggregates not easily resolved by size-exclusion chromatography (SEC).
Viral Vector and Vaccine Purification:
Kalbfuss, B. et al., Journal of Chromatography B, 2007, 849(1–2): 94–104.
Highlight: Demonstrates HIC as an effective method for purifying viral particles while maintaining biological activity.
Peptide and Protein-Drug Conjugates:
Wakankar, A.A. et al., Bioconjugate Chemistry, 2011, 22(3): 449–456.
Highlight: Shows HIC can separate conjugates based on hydrophobic payload loading, similar to ADC DAR analysis.
Biosimilar and Biologic Comparability:
Beck, A. et al., Trends in Analytical Chemistry, 2013, 48: 81–95.
Highlight: Identifies HIC as a tool to detect subtle hydrophobic differences between originator biologics and biosimilars.
Quality Control in Biopharmaceutical Manufacturing:
Shukla, A.A., Thommes, J., Trends in Biotechnology, 2010, 28(5): 253–261.
Highlight: Highlights HIC as a robust and scalable method for monitoring product consistency and impurities in manufacturing.
Enzyme Purification and Characterization:
Geng, X., Wang, F., Journal of Chromatography A, 1993, 640(1–2): 345–351.
Highlight: Demonstrates effective enzyme separation based on surface hydrophobicity while maintaining catalytic activity.
Other Details
The information in this document is subject to change without notice. CellMosaic® assumes no responsibility for any errors or omissions that may appear in this document. Under no circumstances shall CellMosaic be liable, whether in contract, tort, warranty, or under any statute or on other legal theory, for special, incidental, indirect, punitive, multiple, or consequential damages related to or arising from this document, including but not limited to the use of this product thereof.CellMosaic’s products, including but not limited to AqT®, NeIon™, oxLink™ and sxLink™ (collectively, the “Products”), are covered by, or are the subject of, one or more issued patents and pending patent applications, including without limitation: 9,688,663 B2; 8,907,079 B2; 9,511,150 B2; 9,907,854 B2; 9,346,833 B2; 9,518,067 B2; CN104066451B; CN107043339B; CA 2,841,313; EP 2734238 B1; AU2012284055; JP 6240599 B2; and corresponding foreign patents and patent applications. The purchase of the Product conveys solely a limited, non-exclusive, non-transferable, non-sublicensable license to use the Product only for the purchaser’s internal research and development purposes. No right or license is granted, either expressly or by implication, estoppel, or otherwise, under any patent, trademark, copyright, or other intellectual property right of CellMosaic, except as expressly stated herein. Any commercial use of the Product is strictly prohibited without a separate written agreement from CellMosaic. Prohibited commercial uses include, without limitation: (a) the sale, lease, licensing, distribution, or other transfer of the Product or any materials derived from or produced using the Product; (b) the sale, lease, licensing, or other grant of rights to use the Product or any materials derived from or produced using the Product; and (c) the use of the Product to perform services for a fee or other consideration for third parties, including without limitation contract research, screening services, or diagnostic or therapeutic applications. For information regarding commercial licensing or collaboration, please contact us at info@cellmosaic.com.