Product Description
| Download Documents | |
| SDS | Product Sheet |
| COA Lot#_s5.0523 | |
| COA Lot# S8.0919C | |
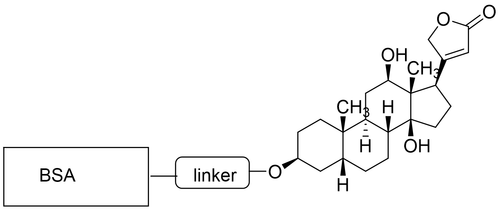
Creatinine is a byproduct of creatine that is produced in the muscles and removed from the body by the kidneys. Measuring the amount of creatinine in the blood and/or urine can predict how well the kidneys are working. Immunoassay is a general method of detecting creatinine. However, since creatinine is a very small molecule it is generally very difficult to generate an antibody that binds strongly to the protein for detection. CellMosaic’s BSA-creatinine conjugate is designed with a flexible linker of optimal length (8-atom length, L8) and a labeling chemistry that does not interfere with creatinine binding. Each lot of the conjugate consists of an optimally loaded BSA (5-10 creatinine molecules per BSA molecule as determined by mass spectrometry or SEC HPLC).
The product is sold as either 1 vial of 1 mg (Cat# CM52117-1MG) or 5 vials of 1 mg (Cat# CM52117-5MG). For bulk orders, please contact us for a quote.
Application
- Suitable for antibody production and creatinine assay development
Key Features of this BSA Creatinine Conjugate
- Frozen liquid in PBS buffer. Ready to use directly after defrosting.
- Long flexible linker and non-interfering labeling chemistry for easy access
- Optimal loading with average of 5-10 creatinine molecules per BSA for strong binding with low background. See each lot for specific loading.
- Concentration accurately determined by UV/HPLC
Example Data
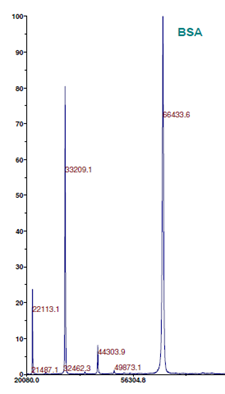
Figure 1: MALDI-TOF MS analysis of BSA (left) and BSA Creatinine L8 (Lot#:s6.0809. Number of Creatinine molecules: 5.7) (right) (Intact MS).
 |
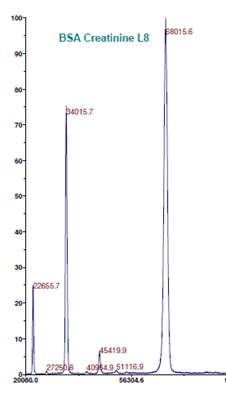 |
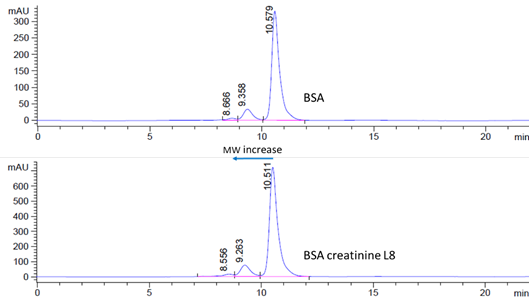
Figure 2: SEC HPLC analysis of unlabeled BSA (top) and BSA creatinine L8 conjugate (bottom, elute earlier. Lot#:s6.0809. Number of Creatinine molecules: 5.7)
Frequently Asked Questions:
If you can’t find the answer you’re looking for or need information on general topics, please visit the main Frequently Asked Questions (FAQs) section.